Overview & Pathophysiology
Periprosthetic osteolysis — progressive bone loss around an arthroplasty implant driven by the biological reaction to wear debris — is the central mechanism of late aseptic loosening in total hip and total knee arthroplasty. It represents a chronic, insidious foreign-body inflammatory response to submicron particles generated at the bearing surface and modular junctions. Understanding the wear particle cascade, the imaging surveillance tools and their limitations, the zone-based radiographic classification systems, and the management decision framework from watchful waiting to prophylactic liner exchange to full revision is essential for the arthroplasty surgeon.
- The wear particle cascade — mechanism of osteolysis: UHMWPE wear particles (0.1–1 µm diameter) generated at the bearing surface are phagocytosed by macrophages and monocytes in the synovial fluid and periprosthetic tissues; activated macrophages release pro-inflammatory cytokines (IL-1β, TNF-α, IL-6, PGE2); cytokines upregulate RANKL (receptor activator of nuclear factor κB ligand) on osteoblasts and stromal cells; RANKL binds RANK on osteoclast precursors, driving their differentiation, activation, and survival; activated osteoclasts resorb bone at the bone-implant or bone-cement interface → osteolytic lesions → progressive implant loosening; NF-κB signalling is the central intracellular pathway; OPG (osteoprotegerin) — the natural decoy receptor for RANKL — is downregulated in the osteolysis environment, further amplifying osteoclast activity
- Particle characteristics: particle size is the most important determinant of biological activity; particles in the 0.1–1 µm range are the most biologically active (readily phagocytosed by macrophages); larger particles (>10 µm) cannot be phagocytosed and stimulate a giant cell reaction instead; particle composition matters — UHMWPE particles are the dominant cause; metal particles (CoCr) from modular junction fretting cause ALTR (adverse local tissue reactions) including pseudotumour; ceramic (alumina/ZTA) particles are the least biologically reactive if they occur
- The effective joint space (Schmalzried concept): wear debris does not affect only the immediately adjacent bone; joint fluid under pressure (during weight-bearing) can migrate along the bone-implant interface, distributing particles to remote areas — the `effective joint space`; this explains why osteolysis can develop at the stem tip, in the greater trochanter, or in the pelvis distant from the acetabular cup; the effective joint space concept explains the distribution of osteolytic lesions that cannot be explained purely by local wear
Radiographic Zone Classification
Zone-based systems allow systematic description and monitoring of periprosthetic radiolucencies on serial radiographs. They are the standard communication tool between radiologists and surgeons, and form the basis for management decisions.
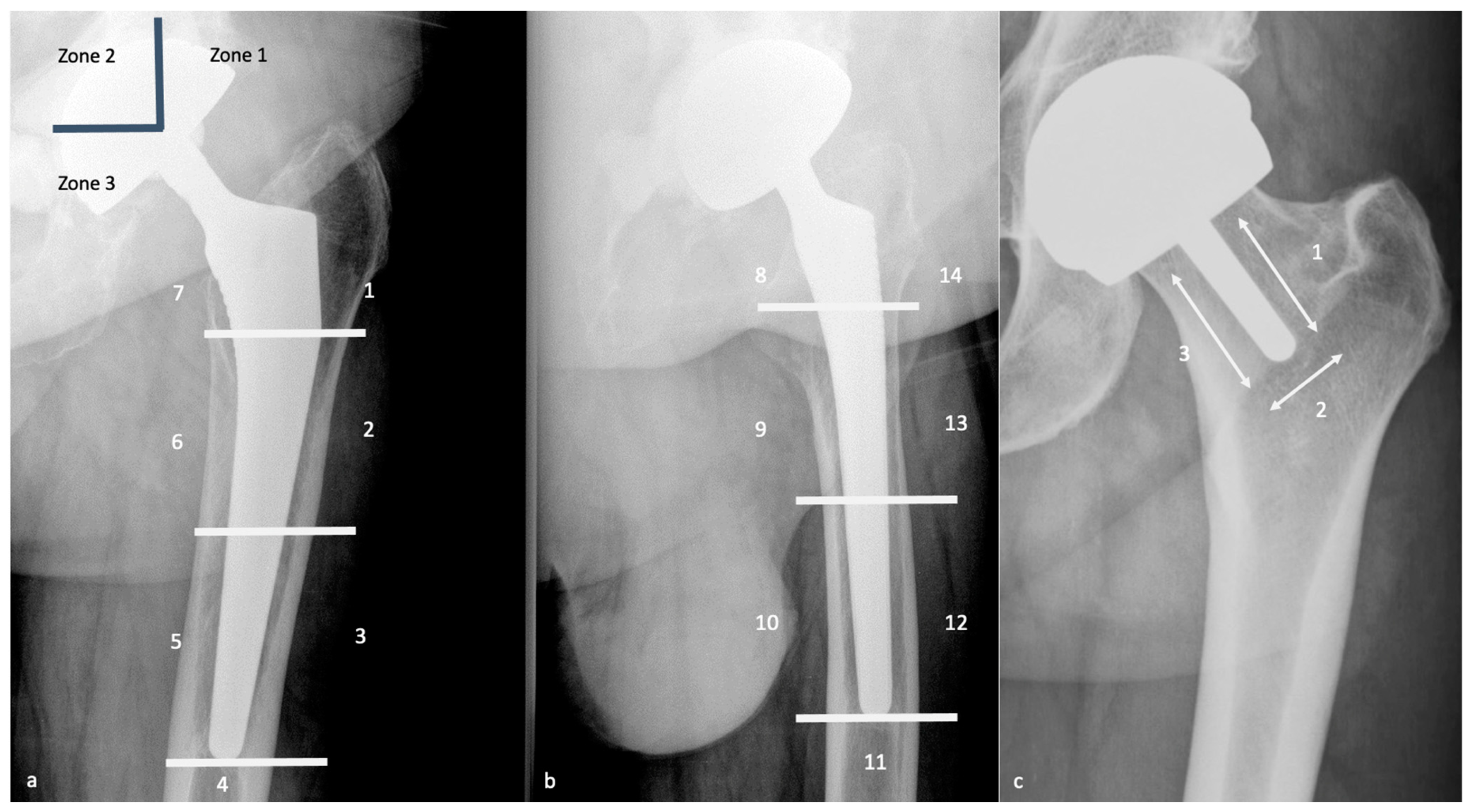
| System | Component | Zones | Clinical Significance |
|---|---|---|---|
| DeLee & Charnley (1976) | Acetabular cup — AP pelvis view | Zone I = superolateral (ilium above cup); Zone II = medial (medial wall); Zone III = inferior (pubis/ischium below cup) | Zone I radiolucency — indicates impending superior cup migration and loosening; Zone II — medial wall deficiency; Zone III — inferior loosening; radiolucent lines >2 mm in any zone = loosening/osteolysis; progressive lines more significant than stable |
| Gruen zones (1979) | Femoral stem — AP view (zones 1–7) + lateral view (zones 8–14) | Zones 1 & 7 = proximal medial/lateral (calcar); Zones 2–6 = diaphyseal (medial/lateral/tip); AP view zones 1–7; lateral view zones 8–14 | Stress shielding (diffuse trabecular thinning) typically affects proximal zones 1 & 7 with distal hypertrophy; osteolysis appears as focal scalloped lucencies; pedestal sign (cortical thickening at stem tip) in zones 4/5 indicates fibrous ingrowth + distal load transfer |
| Radiolucent line threshold | For both cemented and cementless implants: <2 mm stable lucency = acceptable fibrous reaction; >2 mm or PROGRESSIVE lucency = loosening or osteolysis; component migration (measurable subsidence or rotation on serial X-rays) = definitive loosening | Serial comparisons (every 1–2 years) are essential — a single X-ray cannot diagnose progressive osteolysis; compare with earliest available post-operative film as baseline | |
Imaging Modalities
- Plain radiographs — first line but limited: AP pelvis (standing) + lateral hip; DeLee/Charnley and Gruen zones; osteolysis appears as geographic or scalloped radiolucent areas; plain X-rays significantly underestimate the volume and extent of osteolysis due to superimposition of metallic components; one study found plain radiographs detected only 57% of periacetabular osteolytic lesions confirmed by CT (sensitivity 57.6%, specificity 92.9%); serial radiographs every 1–2 years remain the mainstay of arthroplasty surveillance; eccentric positioning of the femoral head within the acetabular cup on AP radiograph = PE liner wear (measurable head penetration)
- CT with metal artefact reduction (MARS CT): gold standard for detecting and quantifying periprosthetic osteolysis; dual-energy CT or metal artefact reduction sequences dramatically reduce scatter from CoCr/Ti implants; 3D volumetric reconstruction allows precise measurement of osteolytic lesion volume and spatial distribution; CT detects osteolysis approximately 2–3 years earlier than plain X-rays; indicated when: (1) plain X-rays suggest osteolysis but extent is unclear for surgical planning; (2) known osteolysis requiring volumetric surveillance; (3) pre-operative planning for revision; acetabular CT is particularly valuable — it assesses integrity of anterior and posterior columns and the posterior wall, which are not well visualised on AP pelvis X-ray
- MARS MRI: superior to CT for soft tissue evaluation — pseudotumours (adverse local tissue reactions from metal debris), ALVAL (aseptic lymphocyte-dominated vasculitis-associated lesions), synovial thickening, fluid collections, and abductor tendon integrity; mandatory investigation for any metal-on-metal THA or trunnionosis with unexplained pain; the Anderson classification of pseudotumours on MRI guides urgency of intervention — Type 1 (benign cystic); Type 2 (mixed cystic-solid); Type 3 (solid — most aggressive, urgent intervention); for polyethylene osteolysis, CT is more useful than MRI; for metallic debris soft tissue reactions, MRI is superior
- Nuclear medicine: Tc-99m MDP bone scintigraphy — increased uptake at sites of accelerated bone remodelling (osteolysis AND stress shielding and infection all show uptake — lacks specificity); labelled leucocyte scan (In-111 or Tc-99m HMPAO) — most specific nuclear medicine test for PJI; 18F-NaF PET/CT — emerging high-sensitivity tool for detecting loosening in patients with equivocal plain radiographs; identifies patients with loose components before X-ray signs are apparent (Ullmark 2020)
- Radiostereometric analysis (RSA): high-precision research tool using tantalum beads implanted at surgery; sub-millimetre measurement of implant migration on serial stereoradiographs; migration >0.2 mm/year in the first 2 years predicts long-term loosening with high specificity; used in implant evaluation trials and phased introduction of new implant designs; not routine clinical practice but increasingly used in national implant monitoring programmes
Intervention Thresholds & Management
- Watchful waiting — surveillance: asymptomatic osteolytic lesions that are small (<5 cm³ on CT), non-progressive, and remote from critical load-bearing zones can be monitored with serial MARS CT (every 12–24 months); surveillance is appropriate when the implant is still well-fixed, the patient is asymptomatic, and the lesion is stable; the aim is to detect progression before structural failure (periprosthetic fracture, cup migration) when intervention is more technically straightforward and outcomes are better
- Critical thresholds for intervention: acetabular osteolysis — lesion volume >5–10 cm³ on CT volumetry, or involvement of >50% of the acetabular dome on any single cross-section, or progressive expansion on serial CT; femoral osteolysis — cortical thinning to <50% of normal cortical width at the stem tip (risk of periprosthetic fracture at that stress riser point); any evidence of component loosening (migration, progressive radiolucent lines) — revision indicated; symptomatic implant
- Prophylactic liner exchange (THA — when acetabular shell is well-fixed): when the shell is well-fixed and in acceptable position but the PE liner is severely worn or perforated → isolated liner exchange + bone grafting of osteolytic cavities through the cup screw holes; technique: remove the worn PE liner; inspect the shell fixation (if mobile → full cup revision required); debride and curette all accessible osteolytic cavities through the existing screw holes (a bone-grafting cannula can be passed through the holes to fill cavities with cancellous allograft); insert a new PE liner of the same or larger femoral head size; if switching to a larger head (improves stability), ensure the new head-neck morse taper is compatible; prophylactic liner exchange before frank loosening produces significantly better outcomes than revision after established loosening
- Full revision arthroplasty: required when the implant is loose; the component is malpositioned (contributing to accelerated wear); osteolysis threatens structural integrity (column fracture, periprosthetic fracture risk); reconstruction follows the Paprosky classification (acetabular defects) or AORI classification (TKA) for bone defect management — see dedicated revision articles
- Medical adjuncts — pharmacological osteolysis treatment: bisphosphonates inhibit osteoclasts (the effector cell of osteolysis); animal and in vitro data are promising; clinical RCT data are limited and show modest effects; not standard of care; denosumab (anti-RANKL monoclonal antibody — directly inhibits the central signalling pathway of osteolysis) is being investigated in clinical trials; theoretically the most targeted pharmacological approach; long-term clinical data awaited; neither bisphosphonates nor denosumab currently has regulatory approval specifically for periprosthetic osteolysis treatment
Wear Reduction Strategies — Prevention
- Highly cross-linked polyethylene (HXLPE): the most important advance in wear reduction; irradiation creates cross-links between PE chains, dramatically improving wear resistance (50–90% reduction in wear rate vs conventional PE in simulator studies); annual head penetration <0.05 mm/year vs 0.1–0.2 mm/year for conventional PE; now standard for acetabular liners in THA; first-generation HXLPE (remelted to quench free radicals) had reduced fracture toughness; second-generation (annealed ± Vitamin E antioxidant stabilisation — e.g., E1, VERILAST) optimises both wear resistance and fatigue properties; HXLPE has dramatically reduced the incidence of clinically significant osteolysis in modern THA cohorts
- Component alignment: cup malposition is the most important modifiable intraoperative factor for bearing wear; cup abduction >55° → edge loading → dramatically increased wear rate (stripe wear on ceramic heads, accelerated PE wear); optimal cup position — abduction 40–45°, anteversion 15–20° (Lewinnek safe zone); robotic and navigated THA improve cup positioning accuracy and reproducibility, reducing outlier rates
- Bearing surface selection: ceramic-on-HXLPE provides the lowest wear rate for MoP bearings (ceramic head is harder and more scratch-resistant than CoCr → less abrasive PE wear); ceramic-on-ceramic provides the lowest overall wear rate but has specific complications (squeaking, liner chipping, catastrophic fracture — rare); metal-on-metal (largely abandoned for THA due to ALTR/pseudotumour); modern HXLPE with CoCr or ceramic heads has excellent wear performance
Exam Pearls
- Wear particle cascade: submicron UHMWPE particles → macrophage phagocytosis → IL-1β/TNF-α → RANKL upregulation → osteoclast activation → bone resorption; NF-κB central pathway; OPG (decoy receptor) downregulated in osteolysis → amplifies osteoclast activity
- Effective joint space (Schmalzried): pressurised joint fluid distributes particles along bone-implant interface to remote areas; explains osteolysis at stem tip, greater trochanter, and pelvic regions distant from the cup
- DeLee & Charnley zones (acetabulum): I (superolateral), II (medial), III (inferior); Zone I radiolucency → impending superior cup migration; >2 mm or progressive lines = loosening/osteolysis
- Gruen zones (femur): 7 zones on AP (1–7) + 7 on lateral (8–14); zones 1 & 7 proximal = stress shielding; pedestal sign at zones 4/5 (stem tip) = fibrous ingrowth + distal load transfer = loosening; focal scalloped lucency = osteolysis
- Plain X-ray sensitivity for osteolysis: only ~57% sensitivity vs CT; specificity 92.9%; underestimates true extent; CT detects 2–3 years earlier; MARS CT = gold standard for volume quantification and surgical planning
- MARS MRI: best for soft tissue — pseudotumour, ALVAL, synovial thickening; mandatory for MoM THA unexplained pain; Anderson classification: Type 1 (cystic), Type 2 (mixed), Type 3 (solid — urgent)
- Intervention threshold: acetabular osteolysis >5–10 cm³ or >50% dome involvement; femoral cortex <50% normal width at stem tip; progressive expansion; component loosening; symptomatic
- Prophylactic liner exchange: well-fixed shell + acceptable position + worn liner; remove liner; curette cavities through screw holes; fill with cancellous allograft; insert new liner ± larger head; pre-loosening intervention = significantly better outcomes than post-loosening revision
- HXLPE: 50–90% wear reduction; <0.05 mm/year; first-generation (remelted) — reduced fracture toughness; second-generation (annealed ± Vit E) — optimised wear AND fatigue; now standard for acetabular liners
- Cup position: abduction >55° → edge loading → dramatically increased wear; optimal 40–45°/15–20°; robotic/navigated THA reduce positioning outliers; most important modifiable intraoperative wear prevention factor
- RSA: tantalum beads; sub-mm migration measurement; migration >0.2 mm/year in first 2 years predicts long-term loosening; research tool; used in implant monitoring programmes; not routine clinical practice
