Overview & Historical Context
Limb salvage surgery (LSS) — the resection of a malignant bone or soft tissue tumour with preservation of the limb — has become the standard of care for the vast majority of primary bone sarcomas of the extremities. Prior to 1970, amputation was the only surgical option for osteosarcoma; five-year survival was below 20% because 80% of patients already had micrometastatic pulmonary disease at presentation. The introduction of effective multi-agent neoadjuvant chemotherapy in the 1970s (high-dose methotrexate, doxorubicin, cisplatin — MAP regimen) transformed the landscape: by sterilising the reactive zone around the tumour and reducing micrometastatic burden before surgery, neoadjuvant chemotherapy enabled safe wide resection margins and dramatically improved five-year survival to 60–80% for localised disease. Simultaneously, advances in implant design, biological reconstruction, and imaging allowed surgeons to replace amputations with limb-sparing procedures in approximately 80–90% of cases at specialist centres.
- LSS vs amputation — oncological equivalence: large studies and a systematic review/meta-analysis have demonstrated no significant difference in five-year overall survival, disease-free survival, or local recurrence rate between LSS and amputation in appropriately selected patients with localised limb osteosarcoma treated with neoadjuvant chemotherapy; LSS does not compromise oncological outcome when adequate surgical margins are achieved; the local recurrence rate after LSS at specialist centres is approximately 5–10%; amputation remains indicated when wide margins cannot be achieved, when major neurovascular structures are encased by tumour, when pathological fracture has seeded the surgical field, or when infection following a previous LSS is uncontrollable
- Most common tumours requiring LSS: osteosarcoma (most common primary malignant bone tumour — peak incidence second decade; distal femur 40%, proximal tibia 20%, proximal humerus 10%); Ewing sarcoma (second most common primary bone sarcoma — broader diaphyseal distribution; also treated with neoadjuvant chemotherapy ± radiotherapy); chondrosarcoma (typically adults; less chemosensitive — surgery is the primary modality); malignant giant cell tumour; metastatic bone disease (selected cases)
Surgical Margins — Enneking Classification
- The Enneking surgical margin system: the gold standard framework for classifying the adequacy of tumour resection; four margin types: (1) Intralesional — the surgical plane passes through the tumour (debulking); leaves macroscopic tumour; unacceptable for malignant tumours; (2) Marginal — the surgical plane passes through the reactive zone (pseudocapsule) surrounding the tumour; microscopic tumour satellite cells may remain; inadequate for high-grade sarcomas but may be acceptable for low-grade tumours or after effective neoadjuvant chemotherapy has sterilised the reactive zone; (3) Wide — the surgical plane passes through normal tissue beyond the reactive zone; a cuff of normal tissue surrounds the entire specimen; the oncological target for LSS of high-grade bone sarcomas; the required `cuff` thickness is debated — historically 2–3 cm; after effective neoadjuvant chemotherapy, smaller margins (<1 cm of normal tissue, or the periosteum/fascia as a barrier) may be acceptable; (4) Radical — the entire muscle compartment containing the tumour is removed en bloc; the highest margin quality but associated with maximal functional loss; rarely required at modern specialist centres
- Margin assessment: intraoperative frozen sections from the bone marrow at the osteotomy site confirm negative medullary margins; the periosteum, fascial planes, and neurovascular adventitia can act as effective barriers even when the measured cuff of normal tissue is small (a 1 mm cuff of periosteum may be oncologically equivalent to a 2 cm cuff of muscle); final margin assessment is confirmed by the pathologist on the resected specimen (permanent histology)
- Neoadjuvant chemotherapy response — Huvos grading: the histological response of the resected tumour to neoadjuvant chemotherapy is the most important prognostic factor after surgical margins; the Huvos grading system (I–IV) is used: Grade I (<50% necrosis — poor response); Grade II (50–89% necrosis — partial response); Grade III (90–99% necrosis — good response); Grade IV (100% necrosis — complete response); Grades III–IV (good response) are associated with significantly better five-year survival (~70–80%) vs Grades I–II (~40–50%); good responders have a sterilised reactive zone, allowing closer margins safely; poor responders may require more radical resection
Reconstruction Options after Tumour Resection
| Method | Description | Advantages | Disadvantages |
|---|---|---|---|
| Endoprosthetic reconstruction (mega-prosthesis) | Modular tumour endoprosthesis replaces the resected bone segment and joint; see dedicated mega-prosthesis article | Immediate stability; early weight-bearing; reliable functional outcome; MSTS 80–87% for distal femur | Mechanical failure over time; infection risk; aseptic loosening; periprosthetic fracture; not ideal for very young children (skeletal immaturity) |
| Allograft reconstruction | Structural (osteoarticular) allograft — cadaveric bone to reconstruct the defect; may be intercalary (diaphyseal) or osteoarticular (including the joint surface) | Biological reconstruction; preserves bone stock; allows ligament reattachment to allograft bone; no implant mechanical failure; suitable for younger patients | Fracture of allograft (15–30%); non-union; infection; subchondral collapse of osteoarticular allograft over time; availability dependent on bone bank; immunological concerns |
| Allograft-prosthesis composite (APC) | A structural allograft provides bone stock while a cemented implant provides the joint surface; combines the bone stock advantage of allograft with the reliable joint function of endoprosthesis | Allows soft tissue (ligament/tendon) reattachment to allograft bone; reliable joint function; suitable for large defects | Technically complex; fracture and non-union of allograft portion; infection risk in immunocompromised chemotherapy patients |
| Vascularised fibula free flap (VFF) | The contralateral fibula (with its vascular pedicle) is harvested and used to bridge the diaphyseal defect; microvascular anastomosis to recipient vessels; the fibula hypertrophies with loading over time | Autologous (no rejection); undergoes stress hypertrophy — eventually becomes a robust bone; no implant; suitable for children (physis may be included); low infection risk | Technically demanding (microsurgery); initial fragility (stress fracture risk); donor site morbidity; long union time; not suitable for periarticular resections without additional reconstruction |
| Extracorporeal irradiation (ECI) and re-implantation | The resected bone segment is irradiated ex vivo (50 Gy single dose) to sterilise any residual tumour cells, then re-implanted and fixed; biological reconstruction using the patient`s own bone | Autologous bone — no immunological issues; preserves exact shape and size; no donor site; cost-effective; MSTS scores 72% (Kamal et al.) | Irradiation weakens the bone (fatigue fracture risk); non-union; delayed incorporation; local recurrence risk if margins were marginal (irradiated bone may harbour residual viable cells if necrosis incomplete) |
| Resection arthrodesis | The joint is fused after tumour resection; the bone defect is bridged with bone graft or intramedullary nail; fusion eliminates joint motion but provides durable stability | Durable; no implant mechanical failure; suitable when soft tissue coverage or extensor mechanism reconstruction is not feasible; useful for proximal tibial resections with poor soft tissue | No joint motion — inferior functional scores (MSTS 68%); non-union; leg length discrepancy; gait adaptation required; limb length must be equalised |
| Rotationplasty (Van Nes) | The distal limb is rotated 180° and reattached; the ankle joint functions as the knee; the limb is fitted with a below-knee prosthesis; biological reconstruction; no implant | Excellent functional outcome (superior to prosthetic for strenuous activity); biological reconstruction — no implant failure; suitable for young children and after failed infected prosthesis | Cosmetically challenging — altered limb appearance; not all patients/families accept it despite superior function; requires specialised prosthetic fitting |
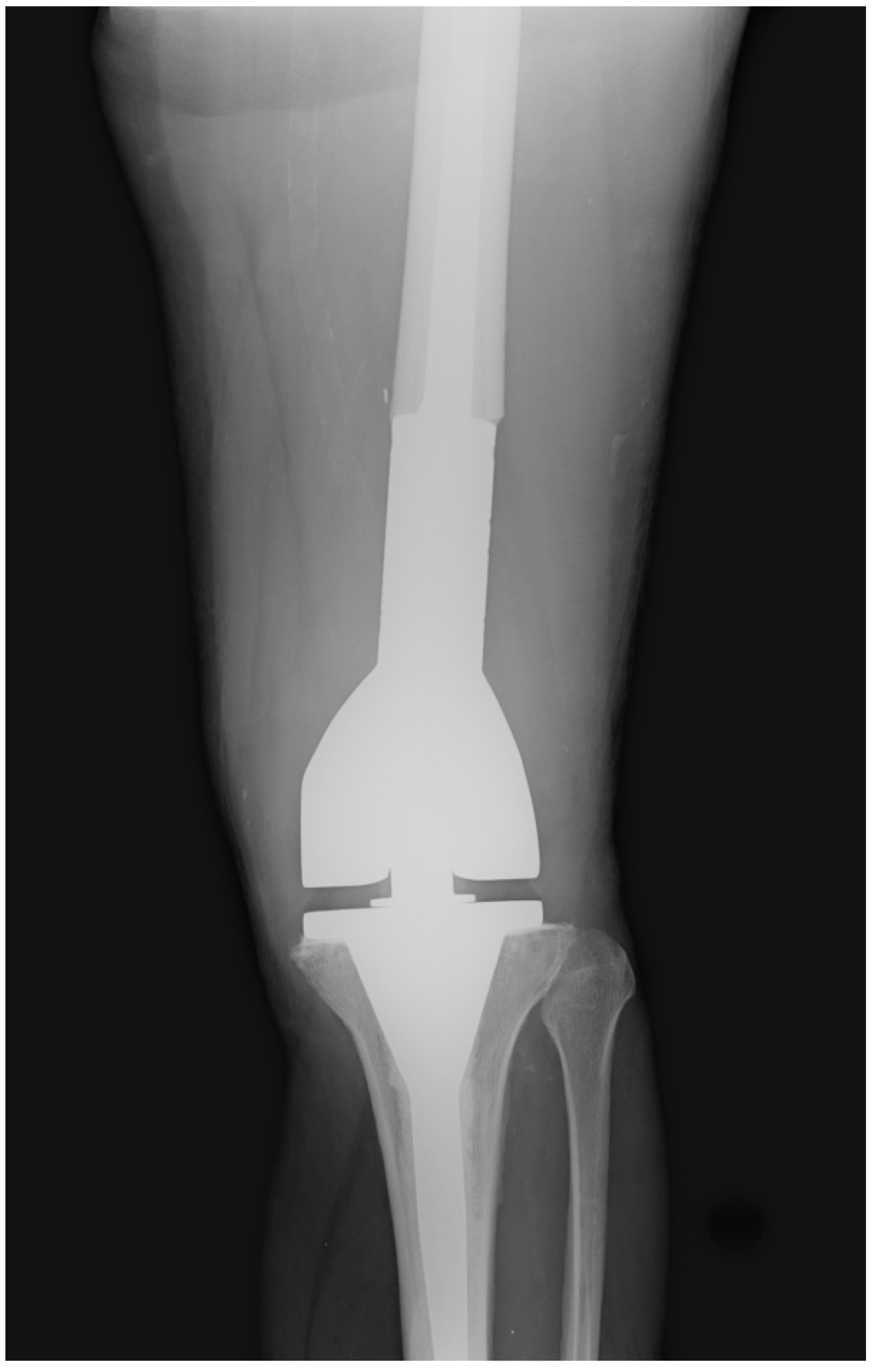
Pre-operative Assessment & Planning
- Staging investigations: plain radiographs of the affected bone (AP and lateral — assess matrix, zone of transition, periosteal reaction, cortical destruction); MRI of the entire bone including the joint above and below (mandatory — defines intramedullary and extraosseous extent, relationship to neurovascular structures, skip lesions, joint involvement — the MRI determines resectability); CT chest (pulmonary metastases — the most common site; present in approximately 15–20% at diagnosis); whole-body bone scan or PET-CT (skip lesions; distant osseous metastases); CT of the primary (cortical detail; relationship to vessels); biopsy (see dedicated biopsy article — must be performed at the treating centre along the planned surgical approach to avoid contaminating a separate skin zone)
- Neurovascular assessment: if the tumour is adjacent to the major neurovascular bundle (popliteal vessels for distal femur/proximal tibia tumours), MRI with vascular sequences assesses displacement vs encasement; if encasement is suspected, CT angiography or MR angiography is performed; encasement of vessels is not an absolute contraindication to LSS — vascular reconstruction (vein graft bypass) can allow limb salvage in selected cases; encasement of the sciatic nerve is a relative contraindication (nerve reconstruction produces poor functional outcomes)
- Biopsy planning: the biopsy must be planned as part of the definitive surgical procedure; the biopsy tract (skin incision, soft tissue path, bone window) must lie within the zone that will be resected at definitive surgery; a misplaced biopsy contaminating a different surgical interval can convert a limb-salvageable tumour into an amputation; the biopsy must be performed at the treating centre — a biopsy performed by a non-specialist surgeon at a referring hospital, placed incorrectly, is one of the most common reasons for preventable amputation in bone sarcoma management
Surgical Principles of Wide Resection
- The `sleeve resection` principle: the quadriceps musculature (vastus intermedius and portions of the other vasti) is routinely sacrificed as part of the sleeve of tissue resected around the distal femoral tumour; this provides a wide margin anteriorly (where the tumour often has anterior soft tissue extension); a sleeve of soft tissue including the vastus intermedius is taken with the specimen; the remaining quadriceps mechanism (vastus medialis, vastus lateralis, rectus femoris) is preserved for extensor mechanism reconstruction; intraoperative frozen sections of muscle margins confirm adequacy of clearance
- Intraoperative navigation: computer-assisted surgical navigation (CAOS) is increasingly used for bone sarcoma resection — CT and MRI images are fused and referenced intraoperatively; the surgeon can visualise the planned osteotomy site in 3D relative to the tumour margins; navigation allows more precise osteotomy placement with confirmed safe margins while preserving maximum bone stock; particularly valuable for joint-sparing resections where the osteotomy must be as close to the tumour as possible while remaining oncologically clear
- Joint-sparing surgery: if the tumour does not involve the growth plate or joint, joint-sparing intercalary resection preserves the physis and both joints (best functional outcome; minimal LLD risk); requires MRI confirmation of adequate tumour-to-physis clearance (>2 cm); intraoperative navigation ensures the osteotomy is safely placed; reconstruction with an intercalary implant (cortical allograft, vascularised fibula, or intercalary endoprosthesis); growing children benefit most from joint-sparing approaches as they avoid the need for expandable prostheses
- Absolute neutrophil count (ANC) before surgery: patients receiving neoadjuvant chemotherapy are immunocompromised perioperatively; surgery must not be performed when the ANC is critically low (<1,500/mm³); the standard protocol is to wait until ANC >1,500 (typically 2–3 weeks after the last chemotherapy cycle) before proceeding with resection to minimise infection risk in a patient who will have a large implant or allograft placed
Functional Outcomes & Complications
- MSTS functional score: the standard outcome tool in orthopaedic oncology (see mega-prosthesis article); LSS with endoprosthesis achieves MSTS 78–87% for distal femur; LSS with ECI autograft 72%; resection arthrodesis 68%; patients adapt remarkably well, especially younger patients; LSS functional outcomes are superior to amputation for most daily activities
- Local recurrence: approximately 5–10% at specialist centres with adequate wide margins; local recurrence after LSS typically requires amputation for salvage; recurrence is influenced by surgical margin quality (most important modifiable factor), tumour grade, Huvos response to chemotherapy, and tumour type; chondrosarcoma (less chemosensitive) and poor Huvos responders have higher local recurrence rates; skip lesions (satellite tumour deposits in the same bone, separated from the main lesion by normal bone — detectable on whole-bone MRI) must be included in the resection specimen or the entire bone must be removed
- Pathological fracture through the tumour: historically considered a contraindication to LSS (fear of seeding the surgical field with tumour cells); however, a meta-analysis (Goulding et al.) demonstrated that LSS in appropriately selected patients with osteosarcoma presenting with pathological fracture does not significantly increase local recurrence or reduce overall survival compared to amputation, when adequate neoadjuvant chemotherapy sterilises the fracture haematoma; pathological fracture is therefore no longer an absolute contraindication to LSS
Exam Pearls
- LSS vs amputation: no significant difference in 5-year OS, DFS, or local recurrence in appropriately selected patients with adequate margins; LSS indicated in ~80–90% of cases at specialist centres; amputation when wide margins unachievable, major NV encasement, or uncontrolled infection
- Enneking margins: Intralesional (through tumour — unacceptable); Marginal (through reactive zone — inadequate for high-grade); Wide (through normal tissue — target for high-grade sarcoma LSS); Radical (entire compartment — rarely needed)
- Huvos grading: histological response to neoadjuvant chemotherapy; Grade III (90–99% necrosis) and Grade IV (100%) = good response; 5-year survival ~70–80%; Grade I–II = poor response; ~40–50%; good response allows safe closer margins
- MAP neoadjuvant chemotherapy: high-dose Methotrexate, doxorubicin (Adriamycin), Cisplatin; 8–12 weeks pre-operatively; sterilises reactive zone + treats micrometastases; transformed 5-year survival from <20% to 60–80%
- Biopsy rule: MUST be performed at treating centre; tract must lie within the planned resection zone; misplaced biopsy contaminating a separate compartment = preventable amputation; commonest avoidable error in bone sarcoma referral pathway
- MRI of the entire bone: mandatory pre-operative investigation; defines intramedullary extent, skip lesions, NV involvement, joint invasion; determines resectability; joint involvement = contraindication to joint-sparing LSS
- ANC >1,500 before surgery: mandatory; wait 2–3 weeks after last chemotherapy cycle; large implant/allograft in immunocompromised patient = catastrophic infection risk if ANC low
- Reconstruction choice hierarchy: mega-prosthesis (most common; immediate stability; best for elderly/non-growing patients); allograft/APC (younger patients; ligament reattachment possible); VFF (children; biological; long-term durability); ECI re-implantation (autologous; cost-effective; fracture risk); arthrodesis (proximal tibia poor soft tissue; sacrifices motion); rotationplasty (children; superior activity levels; cosmetically challenging)
- Joint-sparing surgery: best functional outcome; preserve physis + both joints; requires >2 cm tumour-to-physis clearance on MRI; navigation assists; intercalary reconstruction
- Pathological fracture: no longer absolute contraindication to LSS; meta-analysis shows no significant increase in local recurrence or reduced survival if neoadjuvant chemotherapy achieves adequate sterilisation; decision must be individualised
